An activating point mutation (V617F) of the JAK2-kinase is the molecular hallmark of a group of malignant hematological diseases called chronic myeloproliferative neoplasias (CMN). This group of diseases comprises polycythemia vera (PV), essential thrombocytosis (ET) and primary myelofibrosis (PMF). PV, ET and PMF are characterized by clonal proliferation of myeloid cells and a striking inflammatory syndrome which is the clinical hallmark of the disease in advanced phases. Although high pro-inflammatory cytokine levels have been found in peripheral blood of patients, the molecular basis of this inflammatory response syndrome is poorly understood. Currently, therapeutic options in CMN are limited to symptomatic approaches. In order to develop disease-specific therapies it is of utmost clinical importance and scientific interest to understand the molecular mechanisms of the disease. Therefore, we aim to conduct a comprehensive in vitro and in vivo investigation of the molecular processes - especially the role of β1 and β2 integrin activation in pathophysiology of the disease.
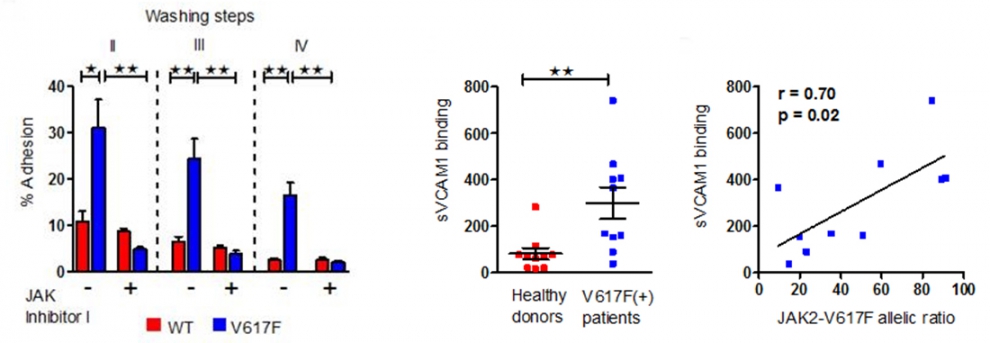
This includes investigation of adhesion of JAK2-V617F positive leukocytes in vitro and in vivo (JAK2-V617F knock-in mouse model). During the previous funding period, we found that JAK2-V617F aberrantly activates β1 and β2 integrins (VLA-4 and LFA-1) on leukocytes in CMN and identified the molecular mechanisms involved. Using the Vav1-Cre x JAK2+/V617F knock-in mouse model established in our laboratory, we aim to dissect the contribution of aberrant integrin activation for the JAK2-V617F- induced cytokine dysregulation, splenomegaly and pro-thrombotic state.